
Absorption spectrum (absorbance vs wavelength) of rutin obtained at the... Download Scientific
Absorbance vs. Transmittance. The absorbance value can range from 0 to infinity, where a higher value indicates a greater absorption of light. Absorbance is commonly used to quantify the concentration of a substance in a solution, as it follows Beer-Lambert's Law, which states that absorbance is directly proportional to the concentration and.

UVvis absorption spectra of absorbance versus wavelength from 200e750... Download Scientific
This page titled 13.1: Transmittance and Absorbance is shared under a CC BY-NC-SA 4.0 license and was authored, remixed, and/or curated by David Harvey. As light passes through a sample, its power decreases as some of it is absorbed. This attenuation of radiation is described quantitatively by two separate, but related terms: transmittance and..
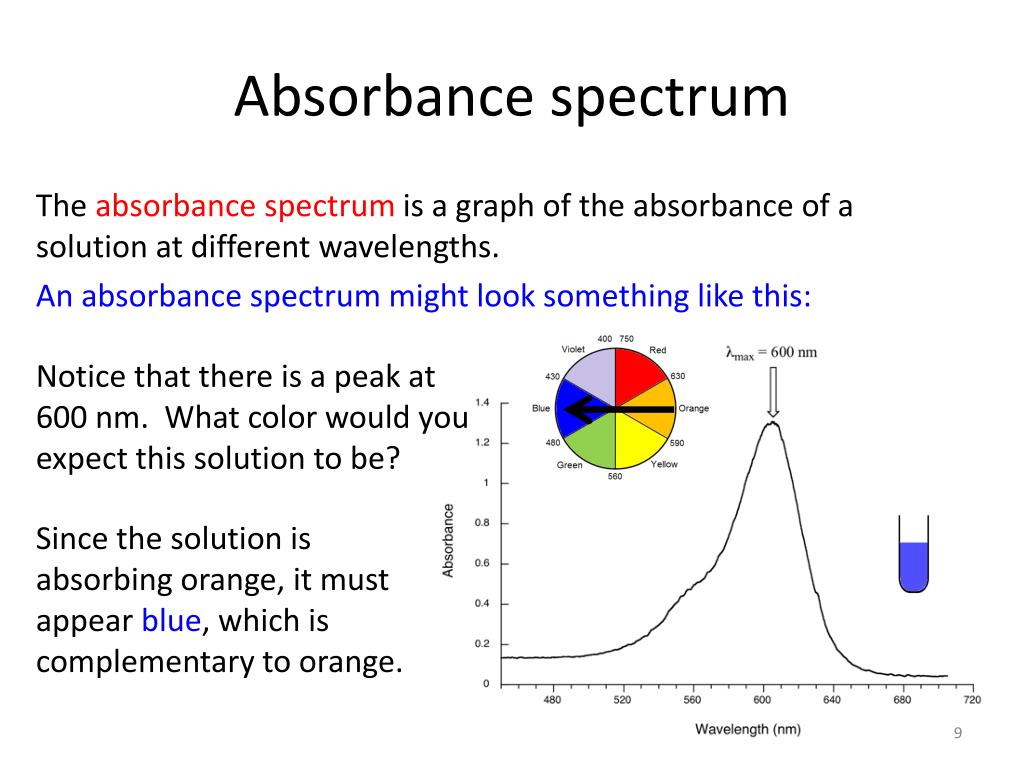
PPT Absorbance spectroscopy PowerPoint Presentation, free download ID3102589
4.4: UV-Visible Spectroscopy. Ultraviolet-visible (UV-vis) spectroscopy is used to obtain the absorbance spectra of a compound in solution or as a solid. What is actually being observed spectroscopically is the absorbance of light energy or electromagnetic radiation, which excites electrons from the ground state to the first singlet excited.
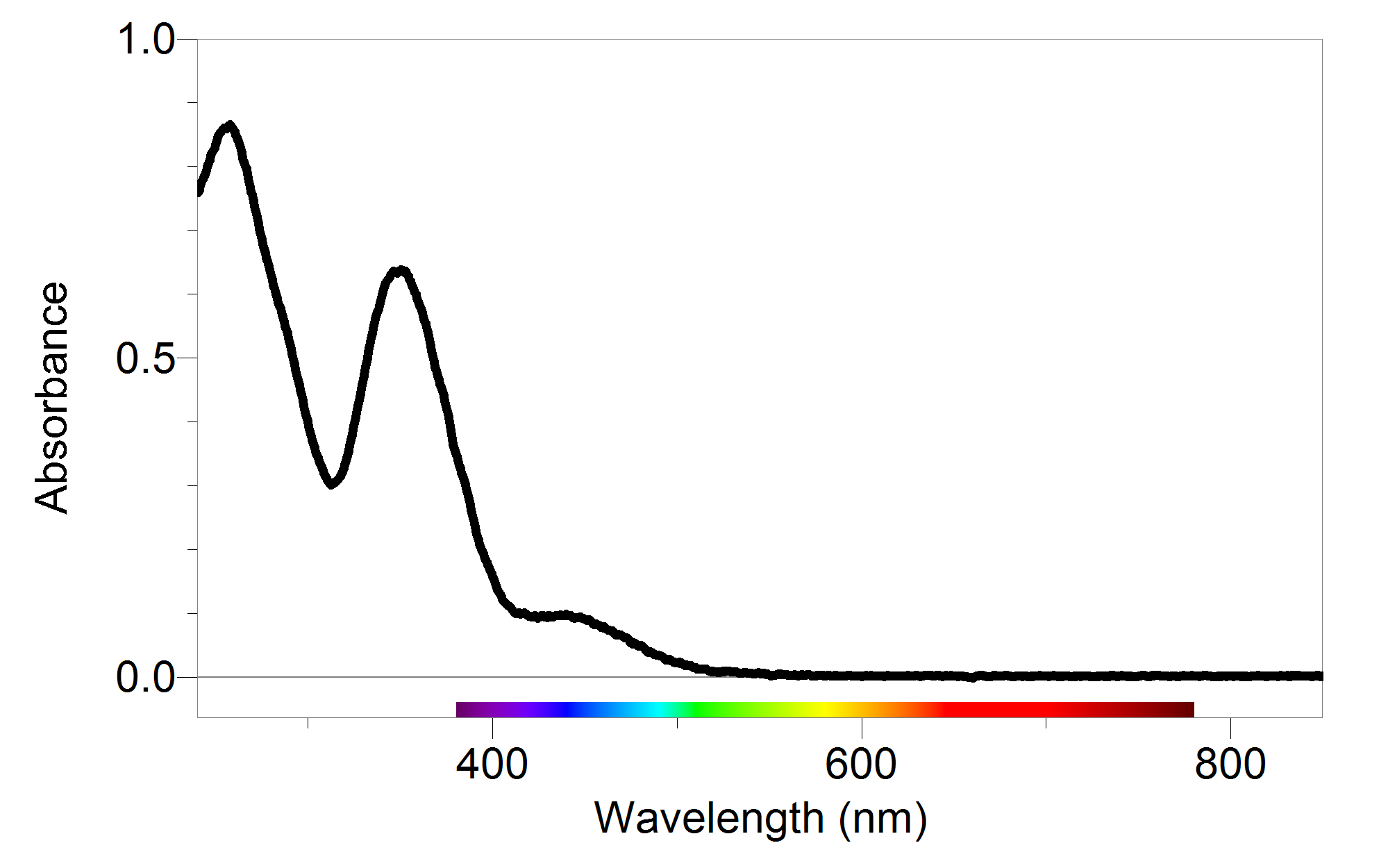
Decoding Your Absorbance Readings Vernier
The act or process of absorbing or the condition of being absorbed. Oct 19, 2021 Absorbance (physics) A logarithmic measure of the amount of light that is absorbed when passing through a substance; the capacity of a substance to absorb light of a given wavelength; optical density. Oct 19, 2021 Absorption A state of mental concentration.

a Absorbance versus wavelength plots of the aqueous solution 4NP and... Download Scientific
The absorbance is directly proportional to the concentration (\(c\)) of the solution of the sample used in the experiment. The absorbance is directly proportional to the length of the light path (\(l\)), which is equal to the width of the cuvette. Assumption one relates the absorbance to concentration and can be expressed as \[A \propto c.

(a) Absorbance vs. Wavelength spectrum and (b) Dependence of absorption... Download Scientific
Absorbance refers to the amount of light absorbed by a substance, while absorption is the process by which a substance takes in another substance or energy. Absorbance is measured using a spectrophotometer, which measures the amount of light absorbed by a substance.
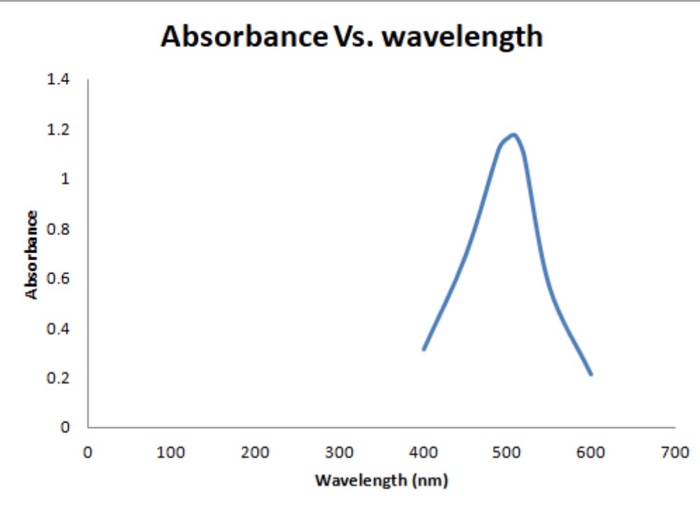
Solved Absorbance Vs. wavelength Absorbance 0 100 200 500
Transmittance is related to absorption by the expression: \(Absorbance (A) = - log(T) = - log(\dfrac{I_t}{I_o})\) Where absorbance stands for the amount of photons that is absorbed. With the amount of absorbance known from the above equation, you can determine the unknown concentration of the sample by using Beer-Lambert Law. Figure 5.

Normalized absorbance and emission intensity spectra versus wavelength... Download Scientific
The result is an absorbance spectrum that shows the intensity of emission as a function of wavelength. As is the case for emission spectra, absorbance spectra range from narrow lines to broad bands. The atomic absorption spectrum for Na is shown in Figure 6.4.4 6.4. 4, and is typical of that found for most atoms.
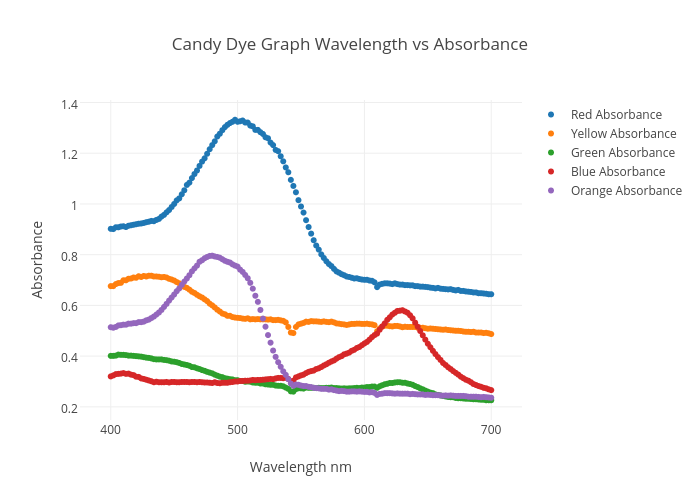
️ Absorbance wavelength graph. Absorbance. 20190218
Absorbance (A) is states how much of the light the sample absorbed. while absorptance is absorbed radiation and incident radiation in a ratio format Cite 1 Recommendation Thomas Mayerhöfer.
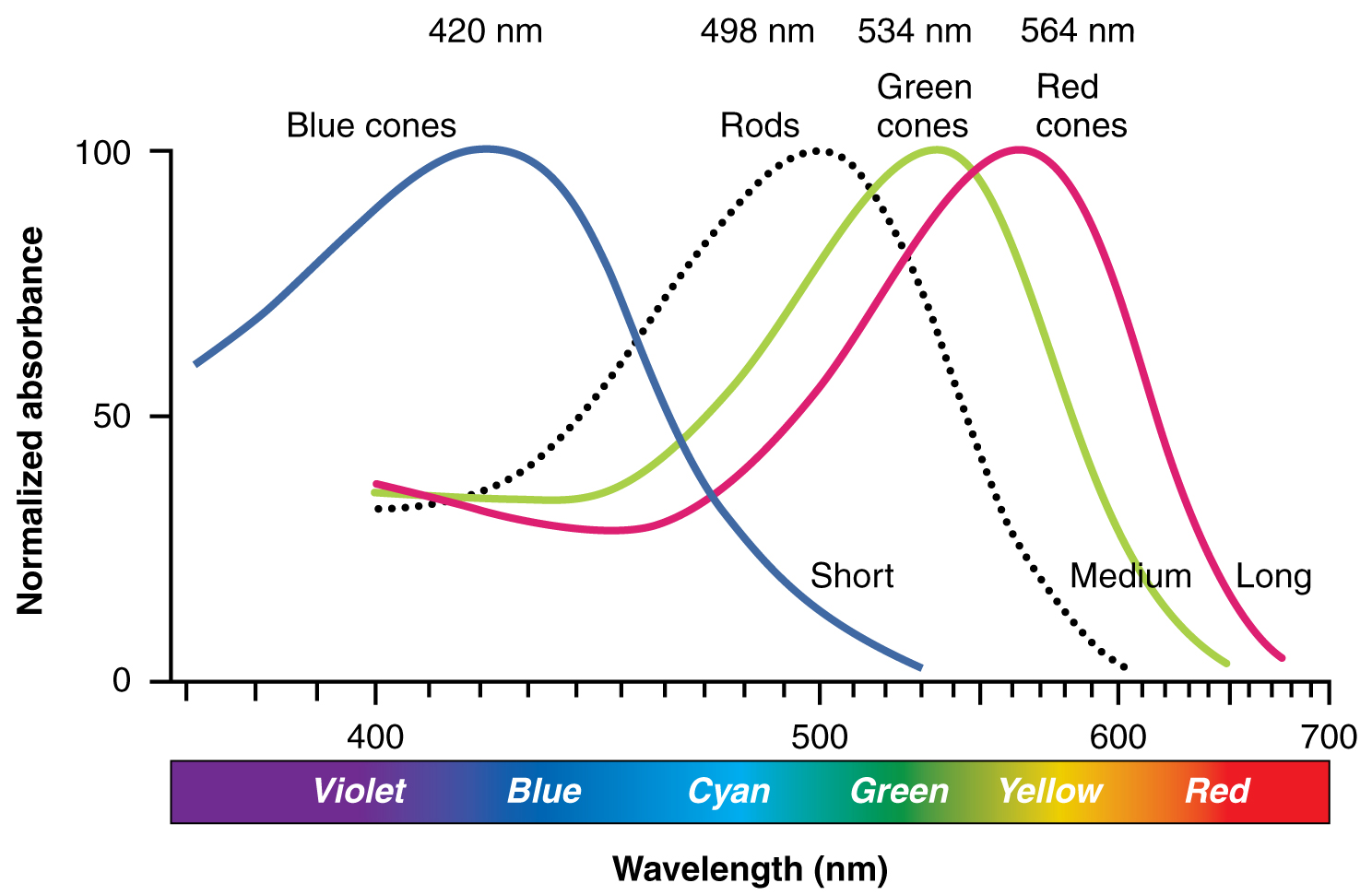
Sensory Perception · Anatomy and Physiology
The main difference between them is that adsorption is the adhesion of particles onto a substance, while absorption involves mass transfer into another material. But, adsorption and absorption involve other differences as well. Here is a comparison of adsorption and absorption, a closer look at their definitions, and examples of each process.

UVvisible absorption spectra of absorbance versus wavelength during... Download Scientific
The term absorption refers to the physical process of absorbing light, while absorbance does not always measure only absorption; it may measure attenuation (of transmitted radiant power) caused by absorption, as well as reflection, scattering, and other physical processes.

Absorbance vs. wavelength graph. Download Scientific Diagram
As nouns the difference between absorption and absorbance is that absorption is the act or process of absorbing or of being absorbed as while absorbance is a logarithmic measure of the amount of light that is absorbed when passing through a substance; the capacity of a substance to absorb light of a given wavelength; optical density.

Absorption Vs Adsorption The Engineering Concepts
Spectrophotometers are a unique set of instruments made to quantitatively measure absorption of visible and ultraviolet (UV-Vis) light or emission of fluorescence compounds. Absorbance approaches directly measure the light absorbed by a sample at a specific wavelength, where the absorbed light measurement is proportional to the concentration of the sample.

Absorption spectra (Absorbance vs wavelength) of 5 NaOCl and dilutions... Download Scientific
Absorption spectroscopy is spectroscopy that involves techniques that measure the absorption of electromagnetic radiation, as a function of frequency or wavelength, due to its interaction with a sample. The sample absorbs energy, i.e., photons, from the radiating field. The intensity of the absorption varies as a function of frequency, and this.

Absorption spectroscopy of haemoglobin species Deranged Physiology
What's the difference between Absorption and Adsorption? Absorption is the process in which a fluid is dissolved by a liquid or a solid (absorbent). Adsorption is the process in which atoms, ions or molecules from a substance (it could be gas, liquid or dissolved solid) adhere to a surface of the adsorbent. Ads.
shows the transmission and absorption spectra of the prepared silicon... Download Scientific
Absorbance is defined as follows: A = -logT For most applications, absorbance values are used since the relationship between absorbance and both concentration and path length is normally linear (as per the Beer Lambert law, described in section 1.9). 1.5 Summary - UV and visible light are part of the electromagnetic spectrum